GONIOTOMY AND CATARACT SURGERY
GONIOTOMY: A SURGERY TO LOWER EYE PRESSURE
When eye drops or lasers are unable to adequately lower eye pressure, goniotomy can be considered. Goniotomy may allow for eye pressure control with or without the use of glaucoma medications.
For patients who qualify for the procedure, goniotomy provides an alternative to bleb-forming glaucoma surgeries (trabeculectomy, Xen) or tube shunts.
How Does Goniotomy Work?
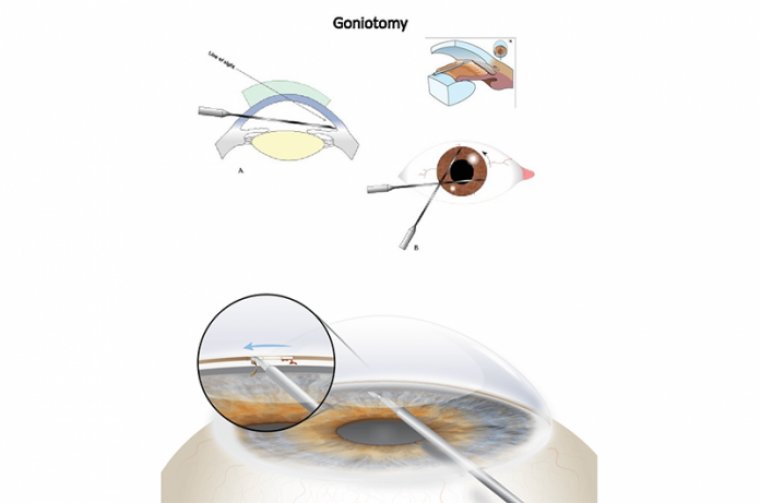
Goniotomy cuts the first layer of the natural drain system (trabecular meshwork). This allows the fluid to leave the eye more easily, and lowers the eye pressure.
The results from a successful goniotomy can provide relief of the intraocular pressure associated with glaucoma, reduced dependency on medication, and help prevent irreversible damage to the optic nerve.
While the Ocular Hypertension Treatment Study (OHTS) provided a guiding principle for patient care, it left ophthalmologists with a lot to think about. The study concluded that lowering intraocular pressure (IOP) with ocular hypotensive medication reduces the risk of developing primary open-angle glaucoma.
It also demonstrated that approximately 90% of patients with elevated IOP may not progress to optic nerve damage and visual field decline. So, for whom do the benefits of treatment outweigh the risks, costs, and inconvenience?
Arriving at the best answer requires consideration of not only IOP and test results, but also a balancing of the efficacy of the various treatment options with life expectancy and quality of life. Even our most efficacious treatment options have side effects.
Topical medications may produce ocular surface disease or hyperemia, which can have effects beyond the cosmetic. For some patients, the treatment burden and cost of topical medications can be too much. The potential adverse effects of other treatment options, while not common, need to be considered as well.
Selective laser trabeculoplasty (SLT) can cause corneal edema, haze, and thinning. Procedures that require a filtering bleb leave patients vulnerable to a long list of potential problems, including susceptibility to trauma and infection.
In our mind, we liken the bleb-creating group of options to fast cars whose brakes don’t necessarily work as well as we’d like. They successfully lower IOP, but have the potential for our hardest-to-control and serious unwanted consequences.
We’re also now familiar with the potential risks of using a device that remains in the eye to facilitate aqueous outflow and lower IOP. Cases of the XEN Gel Stent (Allergan) causing endophthalmitis after eroding through the conjunctiva have been reported.
The more recently introduced microinvasive glaucoma surgery (MIGS) devices aren’t immune to complications, either. The very promising CyPass Micro-Stent (Alcon) was removed from the market because it was shown to cause significant corneal endothelial cell loss.
As we balance treatment safety and efficacy with patients’ quality of life, we must keep in mind that the doctor’s definition of success may differ from the patient’s. Consider, for example, the case of the 62-year-old flight attendant who, despite maximal medical treatment and previous SLT, has IOP in the mid-20s and worsening optic nerve appearance.
The time is right for further intervention, and she undergoes a trabeculectomy. From the surgeon’s perspective, the outcome is great: vision dropped only two lines to 20/40; IOP is 9 mmHg; and visual field and optic nerve are stable.
However, the patient sees the outcome differently. Medications are eliminated, but her vision has declined to 20/40. Given that her job is performed at high altitude and her hobbies are skiing and scuba diving, the thin-walled bleb puts her at risk for eye trauma and endophthalmitis.
This type of situation can deteriorate quickly, from blebitis with good vision to hypopyon and counting-fingers visual acuity. We’ve seen it happen in my practice within a single day.
AN UPTICK IN MIGS, GONIOTOMY
The ongoing quest for efficacious yet ultra-safe glaucoma therapies with as little impact on patients’ lives as possible has definitively manifested in changing treatment patterns.
Data from the Medicare program (1994-2018) track a decline in the number of trabeculectomies performed along with an increase in iStent (Glaukos) trabecular micro-bypass and goniotomy.
Incisional angle surgery has been used for quite some time, including by the surgeons in our group, in the treatment of congenital glaucoma. We’ve published and presented some of our work showing success with trabeculodialysis, i.e., modified goniotomy, in inflammatory glaucoma caused by uveitis.
The technique is different for babies than for adults. In infants, the trabecular meshwork is under tension; therefore, the tissue is easily separated. In uveitic adult eyes, where the tissue isn’t under tension, a wing incision is typically needed to properly excise the tissue.
Performing goniotomy, a relatively noninvasive procedure that can be performed with or without cataract surgery, provided the patient has normal angle anatomy, has been made easier with the availability of the Kahook Dual Blade (KBD).
GONIOTOMY WITH THE KAHOOK DUAL BLADE
Several features of the Kahook Dual Blade maximize safety and make it easier to use than the standard goniotomy blade. A sharp tip facilitates piercing of the trabecular meshwork.
A 30- to 40-degree ramp elevates and stretches the tissue, which allows for a more precise cut. Dual blades at the top of the ramp create parallel incisions that neatly excise a strip of the raised trabecular meshwork.
The blade includes a foot plate/heel that helps the surgeon to follow the curve of the eye and avoid damaging the anterior wall of Schlemm’s canal and sclera.
When performing KDB goniotomy for IOP control in conjunction with cataract surgery, the surgeon—viewing through a gonio-prism—passes the blade through the main incision, across the anterior chamber inflated with viscoelastic, into the nasal iridocorneal angle.
The design of the KDB has enabled the development of three different techniques for its use. With the inside-out technique, the surgeon moves with a forehand to the left and a backhand to the right, which results in two “balled-up” areas of trabecular meshwork on either side that are left in place.
With the outside-in technique, the surgeon moves two to three clock hours to one side, treats to a point across from the clear corneal incision, then reverses course two to three clock hours the other way, which results in a free trabecular meshwork strip.
The third “mark and meet” technique, the surgeon places an initial treatment for one clock hour at the expected site of the treatment’s completion, then with another forehand maneuver moves three to four clock hours to the other side and treats to the area of the initial pass.
This results in a free trabecular meshwork strip that can be removed with forceps. The KDB handles well in the surgeon’s hand regardless of which of the three techniques he or she prefers to use. Having an assistant hold the gonio-lens during the first few cases with the KDB can be helpful.
Other aspects of cataract surgery/KDB goniotomy to consider include:
• Patient selection. Goniotomy is best performed in patients whose iridocorneal angle is sufficiently open and accessible. That said, trypan blue, especially if it’s already being used in cataract surgery, can help to make the trabecular meshwork more visible.
Beyond the requirement of a visible, accessible angle, I’m most comfortable recommending KDB goniotomy for patients who’ve been using one or two IOP-lowering medications and whose optic nerve and visual field are relatively healthy.
Because a postoperative spike in IOP is possible, we don’t recommend the procedure for patients with more advanced disease, i.e., in whom a transient IOP increase constitutes too high a risk for optic nerve damage.
In our experience, the more severe glaucoma, the higher the risk of a post-op IOP spike and the longer it may last. To reduce the risk of postoperative hyphema, it’s helpful to have patients who are on an aspirin regimen discontinue it for a couple of weeks before surgery.
• Pre-op informed consent. Patients must certainly be made aware of the possibility of hyphema after goniotomy and that, as a result, their vision in the immediate post-op period may not be as sharp as if they had undergone only cataract surgery.
• Microscope positioning. As is necessary in iStent placement, obtaining the proper view of the angle during goniotomy requires that the patient’s head be turned away from the surgeon and the microscope be tilted. Good positioning may require the observer microscope to be removed or somehow prevented from falling into the field of view during the procedure.
• Cataract surgery or goniotomy first? Although KDB goniotomy can be done before the accompanying cataract surgery, I (and likely most other surgeons) prefer to perform the cataract surgery first. The viscoelastic can be left in the eye following IOL placement to keep the eye firm for goniotomy.
• Viscoelastic selection. We prefer the eye to be pressurized quite a bit for goniotomy. It’s common practice to use a dispersive viscoelastic, such as Viscoat (Alcon), during phaco and a cohesive viscoelastic, such as ProVisc (Alcon) or Healon (Johnson & Johnson Vision), to expand the capsule for IOL insertion.
That approach would work just fine for cataract surgery plus goniotomy, too. We’ve also had success with Healon GV (Johnson & Johnson Vision)—which is even more cohesive—in KDB goniotomy. The thickness serves as a tamponade against the small amount of bleeding that may occur.
It also provides a bit more “strength” for keeping the anterior chamber deep. Viscoela tic choice can be each surgeon’s preference, but it’s important to keep in mind that when not adequately removed from the eye, the thicker products could increase the risk of an IOP spike.
• Minimal force applied. Even though surgeons are more comfortable having tactile feedback during a procedure, it’s important to not push too hard during KDB goniotomy, especially in initial cases. The blade is surprisingly sharp. If you’re feeling something, you’re likely pushing too hard. All that’s necessary is to gently glide the blade along. The displaced or removed tissue will become visible.
• Postoperative IOP increase. To help prevent an IOP spike following cataract surgery/goniotomy, a drop of beta blocker or alpha-2 agonist can be instilled at the end of the procedure. However, we currently give our patients a Diamox Sequels (Teva Pharmaceuticals) capsule in the recovery area.
The oral medication route is more costeffective than delivering it intravenously and is effective for staving off an IOP increase. We see our patients again the day after surgery. If everything looks good and the IOP is in the mid-teens, additional Diamox shouldn’t be needed.
Alternatively, a judgment call can be made to continue Diamox for a few more days until the eye quiets down and pressure starts responding.
iSTENT VS. KDB GONIOTOMY: RECENT COMPARATIVE STUDIES
Although a large number of head-to-head comparisons of the various MIGS options have yet to be done, two recent comparative studies are of interest.
In a retrospective analysis, the KDB Goniotomy Study Group compared cataract surgery plus KDB excisional goniotomy with cataract surgery plus iStent trabecular micro-bypass in eyes with mild to moderate open-angle glaucoma and visually significant cataract.
The study involved 315 eyes of 230 adults. The KDB+ phaco group included 190 eyes, and the iStent+phaco group included 125 eyes. All were being treated with one or more topical IOP-lowering medications prior to surgery.
Baseline characteristics were similar in both groups except with regard to a few statistically significant differences. Baseline mean IOP (standard error) was significantly higher in the KDB+phaco group [18.2 (0.3) mmHg] than in the iStent+ phaco group [16.7 (0.3) mmHg] (p=0.001).
Also, the KDB+ phaco group was slightly younger on average and included more Hispanic and African-American patients as well as more cases of exfoliation and pigmentary dispersion than the iStent+phaco group.
Only eyes with uncomplicated cataract surgery that were followed for 12 months and didn’t require a subsequent glaucoma procedure were included in the analysis.
In each case, the 13 surgeons who performed the procedures completed the cataract surgery first. During each of the postoperative time points evaluated, the proportion of eyes that achieved IOP reduction ≥20% was higher in the KDB+phaco group than in the iStent+phaco group.
At month 12, 64.2% of the KDB+phaco eyes and 41.6% of the iStent+phaco eyes had IOP reductions ≥20%. The proportion of eyes achieving a reduction in the use of IOPlowering medications postoperatively was comparable in the two groups.
Additionally, at month 12, medication use was reduced by at least one medication in 80.4% of KDB+phaco eyes and 77.4% of iStent+phaco eyes. Adverse events were statistically similar between the two groups of eyes.
They were generally mild to moderate and resolved without treatment. In another study focused on endothelial cell changes, 13 patients with visually significant cataract and mild to moderate open-angle glaucoma underwent cataract surgery plus KDB goniotomy in one eye and cataract surgery plus iStent implantation in the fellow eye.
Baseline endothelial cell density was similar among eyes but decreased significantly more postoperatively in the iStent eyes (-243.2 ± 247.2 cells/mm2) compared with the KDB eyes (-63.1 ± 256.3 cells/mm2).
In other words, a 9.2% reduction in the iStent eyes vs. a 2.6% reduction in the KDB eyes. This reminds us of the potential consequences and dangers of too much time spent in the eye, the level of manipulation of the eye, as well as leaving a device inside. None of which are issues when performing goniotomy with the KDB.
KDB GONIOTOMY: A VALUABLE OPTION FOR PATIENTS, SURGEONS, AND ASCS
The KDB has been a useful addition to my treatment toolbox. It makes goniotomy easier to master than with a standard blade while enhancing intraoperative safety, and goniotomy holds a unique place in the glaucoma surgery spectrum.
It’s much less extensive compared with trabeculectomy or other procedures that necessitate a filtering bleb, and it leaves no device inside the eye to potentially create problems. In my hands it has been quite predictable—a welcome attribute—in lowering IOP and often reducing medication use.
The predictability is key, especially in younger glaucoma patients for whom I need to provide years and years of quality vision and want to do so with as little effect on their quality of life as possible. It’s worth mentioning, too, that our ASC manager appreciates the KDB’s efficiency with regard to surgeon time and its lower cost compared with MIGS implants.